How do lasers work?
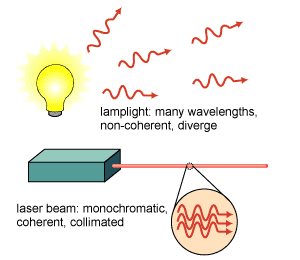
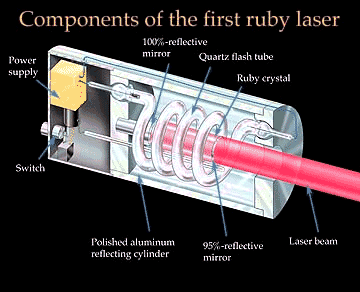
Packets of light energy are called photons; this energy is quantized (van Kessel 171). In a laser beam, photons in the light travel precisely one behind the other; this creates consistent waves that are in phase which causes the beam to seem like one big continuous wave (van Kessel 204). Furthermore, the waves in a laser beam are all perfectly parallel to each other. This results in almost no light being diverged/spread out while traveling (van Kessel 204). Located within the laser is a cylinder that is packed with either gases or solid crystal. Common substances used are helium, neon, and ruby (Farndon).
LASER stands for Light Amplification by Stimulated Emission of Radiation (van Kessel 203). To explain how lasers work, principles of quantum mechanics are used. Quantum mechanics is a theory which uses the wave properties of electrons in an atom to explain atomic structure (van Kessel 199). The quantum revolution was first started by Planck. In 1900, he came to the conclusion that light is released in bursts of energy when atoms are heated; thus, the energy released is not continuous (van Kessel 169). In 1971, Albert Einstein first predicted that for an electron to get to a higher energy level, a certain amount of energy/ photon is needed. Using this, the laser theory explained that if the exact photon hits the atom while it is already in its excited phase, the electron would fall back to a lower energy level (van Kessel 204). At the same time that the electron goes back to a lower energy level, another photon will simultaneously be released. That photon will have the same energy as the initial photon that first hit the excited electron and will also travel in the same direction as it (van Kessel 204).
Thus, if electrons in numerous atoms were brought to an excited state, a chain reaction could occur with just one photon. When the photon hits one electron, two photons will now be released and when the two photons hit the next atom, four photons will now be released (van Kessel 204). One photon can cause a whole beam of photons; for this reason the LASER acronym includes the word “amplification” (van Kessel 204).
LASER stands for Light Amplification by Stimulated Emission of Radiation (van Kessel 203). To explain how lasers work, principles of quantum mechanics are used. Quantum mechanics is a theory which uses the wave properties of electrons in an atom to explain atomic structure (van Kessel 199). The quantum revolution was first started by Planck. In 1900, he came to the conclusion that light is released in bursts of energy when atoms are heated; thus, the energy released is not continuous (van Kessel 169). In 1971, Albert Einstein first predicted that for an electron to get to a higher energy level, a certain amount of energy/ photon is needed. Using this, the laser theory explained that if the exact photon hits the atom while it is already in its excited phase, the electron would fall back to a lower energy level (van Kessel 204). At the same time that the electron goes back to a lower energy level, another photon will simultaneously be released. That photon will have the same energy as the initial photon that first hit the excited electron and will also travel in the same direction as it (van Kessel 204).
Thus, if electrons in numerous atoms were brought to an excited state, a chain reaction could occur with just one photon. When the photon hits one electron, two photons will now be released and when the two photons hit the next atom, four photons will now be released (van Kessel 204). One photon can cause a whole beam of photons; for this reason the LASER acronym includes the word “amplification” (van Kessel 204).